World
World Ozone Day 2020: Interesting Ozone Layer Facts

International Day for the Preservation of the Ozone Layer, also known as World Ozone Day or World Ozone Layer Day, is praised every year on September 16 to raise awareness about ways that are effective in protecting the ozone layer. The theme or slogan for World Ozone Day 2020 is ‘Ozone for life: 35 years of ozone layer protection’. Here are some interesting facts about the ozone layer.
Theme of World Ozone Day 2020
The current year’s theme for the World Ozone Day 2020 is “Ozone for life: 35 years of ozone layer protection”.
In 1985, the governments at the global level received the Vienna Convention for the Protection of the Ozone Layer. After two years in 1987, the Montreal Protocol was drafted where the governments, researchers, and industry cooperated to remove 99 percent of all ozone-depleting substances.
As per the United Nations Environment Program (UNEP), the ozone layer is recouping and expected to return to pre-1980 values by mid-century all thanks to the Montreal Protocol. Then, supporting the Montreal Protocol, the Kigali Amendment, which came into force in 2019, will progress in the direction of lessening hydrofluorocarbon (HFCs) and greenhouse gases that have climate-warming potential and are harming to the environment.
What is ozone?
Ozone is a naturally happening molecule. An ozone molecule is comprised of three oxygen atoms. It has the chemical formula O3.
Ozone is a gas comprised of three oxygen atoms (O3). It happens naturally in small (trace) amounts in the upper atmosphere (the stratosphere). Ozone protects life on Earth from the Sun’s ultraviolet (UV) radiation. In the lower atmosphere (the troposphere) close to the Earth’s surface, ozone is made by chemical reactions between air pollutants from vehicle exhaust, gasoline vapors, and other emissions. At ground level, high concentrations of ozone are harmful to individuals and plants.
What is the ozone layer?
The ozone layer is the basic term for the high concentration of ozone that is found in the stratosphere around 15–30km above the earth’s surface. It covers the whole planet and protects life on earth by engrossing hurtful ultraviolet-B (UV-B) radiation from the sun.
Why is UV-B radiation bad?
Delayed exposure to UV-B radiation is connected to skin cancer, genetic harm, and immune system suppression in people and animals, and lower-yielding crops.
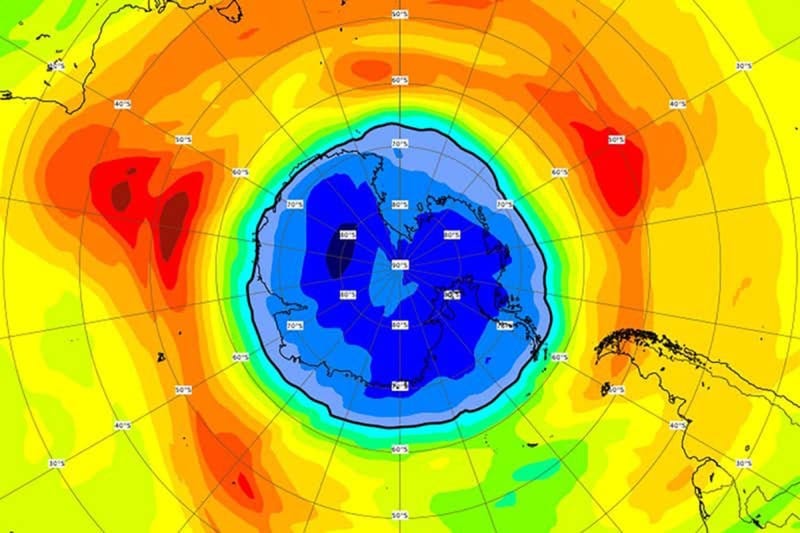
What is the ozone hole?
Chemicals containing chlorine and bromine atoms are released to the atmosphere through human activities. These chemicals consolidate with certain weather conditions to cause responses in the ozone layer, prompting ozone molecules to be crushed. The depletion of the ozone layer happens around the world, be that as it may, the serious depletion of the ozone layer over the Antarctic is regularly alluded to as the ‘ozone hole’. Expanded depletion has recently begun happening once again the Arctic also.
Ozone Layer Facts
The ozone layer is one of the layers in the earth’s atmosphere, contained generally within the stratosphere’s layer, at a distance of somewhere in the range of 15 and 30 km from the surface of the earth. Ozone is a molecule, with a strong smell and it is blue. In 10 million air molecules, just 3 are ozone, making it substantially less common than different molecules, for example, oxygen, which makes up around 2 million of each 10 million air molecules.
Regardless of the generally small percentage of ozone molecules in the earth’s atmosphere, it is critical for life on earth. Ozone molecules assimilate the UVB ultraviolet light that accomplishes such great deal harm on the planet. Ozone assists with protecting individuals, animals, plants, and marine life from the sun’s harmful rays.
Interesting Ozone Layer Facts
- The chemical formula for ozone is O3. That implies that it is comprised of three oxygen molecules bonded together. Thus, it is otherwise called ‘trioxygen’ where ‘tri’ signifies three.
- The ozone layer was found by Charles Fabry and Henri Buisson, French physicists, in 1913.
- The British meteorologist G.M.B. Dobson built up a spectrophotometer and set up ozone monitoring stations the world over somewhere in the range of 1928 and 1958. His work around there prompted the naming of the ‘Dobson unit’, which alludes to a measure of ozone overhead.
- Ozone has a color: it is pale blue.
- The ozone layer can retain up to 98% of the sun’s ultraviolet light, and 90% of the ozone is situated in the stratosphere layer of the atmosphere.
- You may have smelled ozone without acknowledging it. If, after a helping storm you smelled a pungent scent in the air, at that point this may well have been ozone which (just as existing in the ozone layer over our planet) can be produced in tiny quantities when lightning strikes.
- The ozone layer is a layer of ozone that exists in the upper atmosphere of our planet earth. This layer of the atmosphere is likewise called the stratosphere.
- The ozone layer encourages us by filtering the destructive UV rays of the sun out of the atmosphere before they arrive at earth. This assists with guarding us against the ailments and diseases that exposure to UV rays can cause, for example, skin cancers and cataracts.
- The ozone layer has a thickness of 20 km.
- The UVB radiation that is screened out by the ozone layer can cause health problems when exposure is excessively high, which is the reason the ozone layer is crucial to human and animal survival.
- UVB radiation can cause immune system suppression, cataracts, genetic damage, and even skin cancer.
- Albeit high concentrations of UVB exposure can be hazardous, some are fundamental for the production of vitamin D, which is likewise vital for human health.
- CFCs, which were chemicals that contain chlorine, was used in aerosols, coolants, and different manufacturing processes. CFCs caused a hole in the ozone layer to form. This is because a single chlorine atom can annihilate 10s of thousands of ozone molecules.
- The ozone layer has been harmed by substances known as chlorofluorocarbons (CFCs) which were used in a variety of products, for example, protecting foam, solvents, and refrigerants. The molecules in CFCs destroy ozone molecules at a rate of 1:100,000 which makes them perilous to the ozone layer.
- A moderate yet nevertheless significant exertion has been made worldwide to boycott or limit the use of CFCs trying to stop the decimation of the ozone layer.
- On the off chance that individuals quit producing ozone-destroying substances, the ozone layer might be equipped for recuperating by 2050.
- A few nations have prohibited the use of CFC aerosol sprays altogether, for example, Canada, Norway, and the U.S. different applications of CFCs were still permitted notwithstanding.
- At the point when CFCs were prohibited, the hole in the ozone layer started to heal, an incredible demonstration of how when individuals settle on the correct decisions we can mend the harm that we have done to the environment.
- Ozone is significantly less steady than the oxygen in the air that we breathe, which makes the ozone layer truly sensitive and more inclined to separate.
- A hole in the ozone layer will likewise make earth’s temperatures rise drastically.
- A hole in the ozone was found in the Antarctic in 1985. It’s not an actual hole as several individuals accept, yet it alludes to lost 60% of the ozone that is generally present from September to November every year.
- The ozone layer is gaseous since it has an extremely low boiling point: for example less 112 degrees celsius. The boiling point of any substance is where it abandons a liquid into a gas.
- The ozone layer is somewhere in the range of 10 and 50 km above the earth, contingent upon which locations you are estimating from.
- Even though the ozone layer in the stratosphere assists with ensuring our health and our planet, on the off chance that we took in pure ozone in enormous amounts we would die as we would discover it exceptionally poisonous to our respiratory system.
- Our car’s exhaust pipes and different types of machinery that use gasoline produce ozone as toxic fumes mingle with other chemical compounds. This is another case of how ozone can be poisonous.
- In different regions around the world, the ozone layer has been depleted by about 20%.
- The ozone layer in the stratosphere isn’t made up exclusively of O3 ozone gases. Ozone conforms to 0.0015% of the stratosphere: nevertheless, this is sufficient to give our planet the protection that we need.
- The ozone layer weighs three billion metric tons.
- The sun assists with making ozone. It does as such by taking the O2 oxygen molecules in the air that we breathe (which are comprised of two oxygen molecules bonded together) and using its rays to separate these molecules into two single oxygen molecules which glide freely around. These free-floating molecules may bond together in a couple again to make another breathable O2 oxygen molecule, or they may bond as a group of three to make an O3 ozone molecule.
- Ozone is likewise toxic to plants – another reason why it should remain generally in the stratosphere.
- The Montreal Protocol, ordered in 1989, was set up to attempt to protect the ozone layer by eliminating, around the world, substances that deplete the ozone layer. It has been having any kind of effect and the ozone layer is required to recoup.
- The Montreal protocol is fundamentally an international treaty that is gotten ready for the protection of the ozone layer by decreasing the production of substances that are answerable for ozone layer depletion.
- The ozone was found by a researcher by Christian Friedrich Schoenbein. He made the revelation in 1840.
- The word ozone originates from the Greek word ‘ozein’ which intends to smell. This name is concerning the ozone’s broadly pungent smell. It was Schoenbein, the researcher who found ozone, who decided to give it its name: it is fascinating that he decided to name it after its smell instead of (for example) its color anyway a great many people who have smelt ozone do say that it has an exceptionally particular and memorable scent. It has been contrasted with the smell of rotting seaweed – for sure, numerous individuals have felt that the seaside is loaded up with new ozone enhanced air when in certainty what they are smelling is the seaweed marginally rotting at their feet!
- Ozone has likewise been used in medication. In World War I, for instance, ozone was used to attempt to treat a variety of ailments including trench foot and gangrene. It was additionally used to attempt to purify wounds sustained by soldiers on the battlefield.
- A significant hole in the ozone layer was found above Antarctica in 1985, by researchers. It was immediately settled that CFCs were the reason for this hole and this prompted them to become banned substances.
- September 16th every year is the International Day For The Protection Of The Ozone. It was the UN that began this annual commemorative event to remind us exactly how significant the ozone is.
- On September 16, 1987, a document called the Montreal Protocol was agreed upon. This document bound countries to not release unsafe CFCs into the atmosphere, to ensure the ozone layer. This is the reason the date of September 16th was picked for the annual day committed to the ozone.
- Vienna Convention and Montreal protocol became the first treaties in UN history to accomplish universal ratification on September 16, 2009.
-

 Entertainment3 weeks ago
Entertainment3 weeks agoFull List of Performers and Presenters for the 2026 American Music Awards
-

 Sports4 weeks ago
Sports4 weeks ago2026 FIFA World Cup – Full Schedule, Fixtures, Match Format, Teams, Groups, and Everything You Need to Know
-

 Science4 weeks ago
Science4 weeks agoTotal Solar Eclipse 2026 and 2027: Which Upcoming Eclipse Is Better
-

 Fashion3 weeks ago
Fashion3 weeks agoSelf-Funded and Scaling: Inside the Rise of Fleur Couture
-

 Apps4 weeks ago
Apps4 weeks agoSpotify Honors API Heritage Month 2026 With Playlists, Podcasts, and Artist Support
-

 Apps4 weeks ago
Apps4 weeks agoSpotify Celebrates 20 Years With a New Feature Highlighting Your Complete Listening History
-

 Festivals & Events4 weeks ago
Festivals & Events4 weeks agoInternational Day of Light: History and Significance of the Day
-

 Science4 weeks ago
Science4 weeks agoThings you Should Need to Know about Total Solar Eclipse 2026